MicroRNA-222 regulates melanoma plasticity
Published:
M. C. Lionetti, F. Cola, O. Chepizhko, M. R. Fumagalli, F. Font-Clos, R. Ravasio et al., J. Clin. Med.
Download PDF here
Link to journal
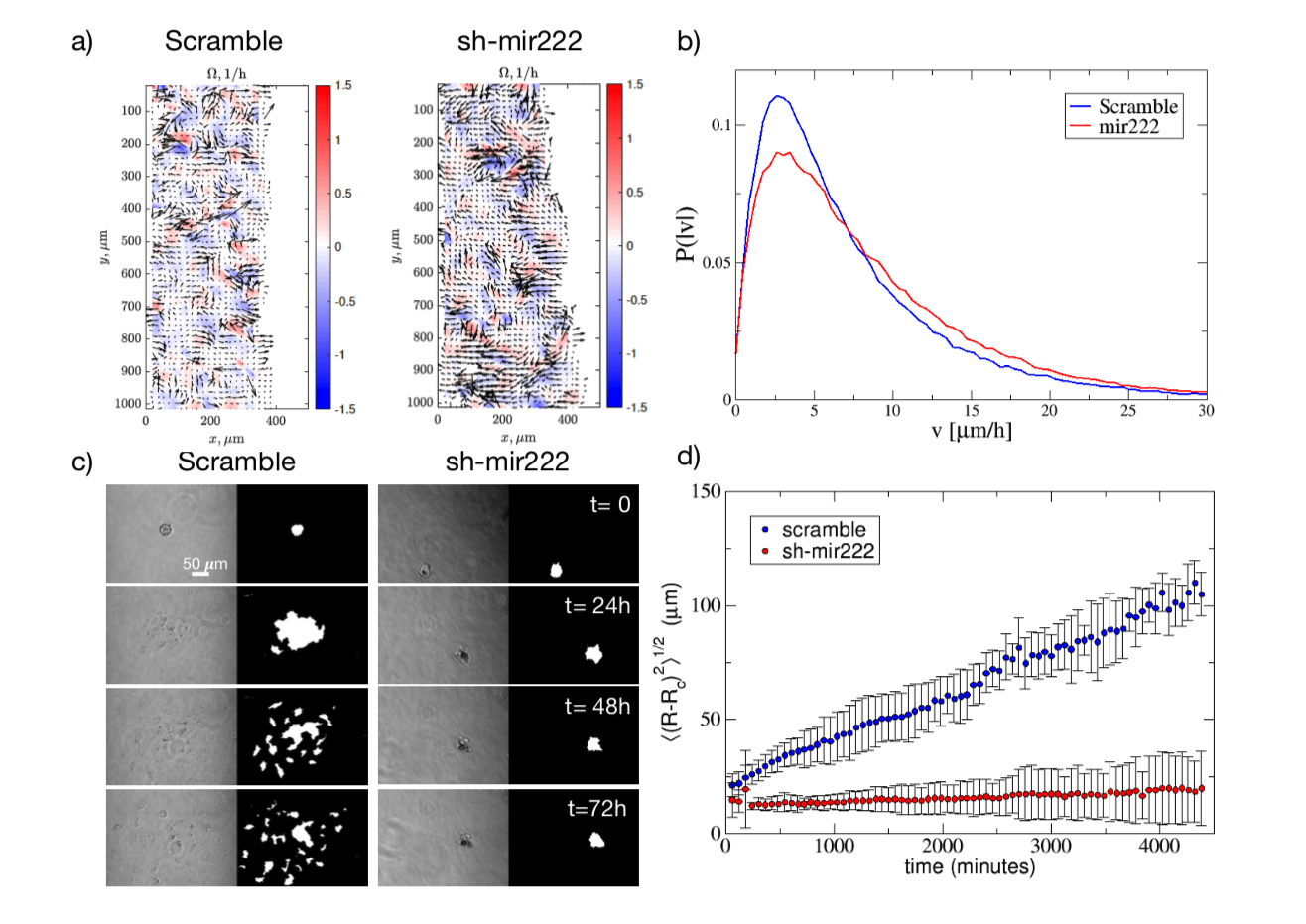
Abstract: Melanoma is one of the most aggressive and highly resistant tumors. Cell plasticity in melanoma is one of the main culprits behind its metastatic capabilities. The detailed molecular mechanisms controlling melanoma plasticity are still not completely understood. Here we combine mathematical models of phenotypic switching with experiments on IgR39 human melanoma cells to identify possible key targets to impair phenotypic switching. Our mathematical model shows that a cancer stem cell subpopulation within the tumor prevents phenotypic switching of the other cancer cells. Experiments reveal that hsa-mir-222 is a key factor enabling this process. Our results shed new light on melanoma plasticity, providing a potential target and guidance for therapeutic studies.
